Grab-AD: Generalizability and Reproducibility of Altered Brain Activity and Diagnostic Classification in Alzheimer's Disease
Dan Jin1,2 Pan Wang3 Andrew Zalesky4,5 Bing Liu1,2,6 Chengyuan Song7 Dawei Wang8 Kaibin Xu1 Hongwei Yang9 Zengqiang Zhang10 Hongxiang Yao11 Bo Zhou12 Tong Han13 Nianming Zuo1,2 Ying Han14,15,16,17 Jie Lu9 Qing Wang8 Chunshui Yu18 Xinqing Zhang14 Xi Zhang12 Tianzi Jiang1,2,6 Yuying Zhou3 Yong Liu1,2,6
1Brainnetome Center & National Laboratory of Pattern Recognition, Institute of Automation, Chinese Academy of Sciences, Beijing, China
2School of Artificial Intelligence, University of Chinese Academy of Sciences, Beijing, China
3Department of Neurology, Tianjin Huanhu Hospital, Tianjin University, Tianjin, China
4Melbourne Neuropsychiatry Centre, Department of Psychiatry, University of Melbourne and Melbourne Health, Melbourne, Victoria, Australia
5Department of Biomedical Engineering, University of Melbourne, Melbourne, Victoria, Australia
6Center for Excellence in Brain Science and Intelligence Technology, Institute of Automation, Chinese Academy of Sciences, Beijing, China
7Department of Neurology, Qilu Hospital of Shandong University, Ji'nan, China
8Department of Radiology, Qilu Hospital of Shandong University, Ji'nan, China
9Department of Radiology, Xuanwu Hospital of Capital Medical University, Beijing, China
10Branch of Chinese PLA General Hospital, Sanya, China
11Department of Radiology, the Second Medical Centre, National Clinical Research Centre for Geriatric Diseases, Chinese PLA General Hospital, Beijing, China
12Department of Neurology, the Second Medical Centre, National Clinical Research Centre for Geriatric Diseases, Chinese PLA General Hospital, Beijing, China
13Department of Radiology, Tianjin Huanhu Hospital, Tianjin, China
14Department of Neurology, Xuanwu Hospital of Capital Medical University, Beijing, China
15Beijing Institute of Geriatrics, Beijing, China
16National Clinical Research Center for Geriatric Disorders, Beijing, China
17Center of Alzheimer's Disease, Beijing Institute for Brain Disorders, Beijing, China
18Department of Radiology, Tianjin Medical University General Hospital, Tianjin, China
Abstract
Alzheimer's disease (AD) is associated with disruptions in brain activity and networks. However, there is substantial inconsistency among studies that have investigated functional brain alterations in AD; such contradictions have hindered efforts to elucidate the core disease mechanisms. In this study, we aim to comprehensively characterize AD-associated functional brain alterations using one of the world's largest resting-state functional MRI (fMRI) biobank for the disorder. The biobank includes fMRI data from six neuroimaging centers, with a total of 252 AD patients, 221 mild cognitive impairment (MCI) patients and 215 healthy comparison individuals. Metaanalytic techniques were used to unveil reliable differences in brain function among the three groups. Relative to the healthy comparison group, AD was associated with significantly reduced functional connectivity and local activity in the default-mode network, basal ganglia and cingulate gyrus, along with increased connectivity or local activity in the prefrontal lobe and hippocampus (p < .05, Bonferroni corrected). Moreover, these functional alterations were significantly correlated with the degree of cognitive impairment (AD and MCI groups) and amyloid-β burden. Machine learning models were trained to recognize key fMRI features to predict individual diagnostic status and clinical score. Leave-one-site-out cross-validation established that diagnostic status (mean area under the receiver operating characteristic curve: 0.85) and clinical score (mean correlation coefficient between predicted and actual Mini-Mental State Examination scores: 0.56, p < .0001) could be predicted with high accuracy. Collectively, our findings highlight the potential for a reproducible and generalizable functional brain imaging biomarker to aid the early diagnosis of AD and track its progression.
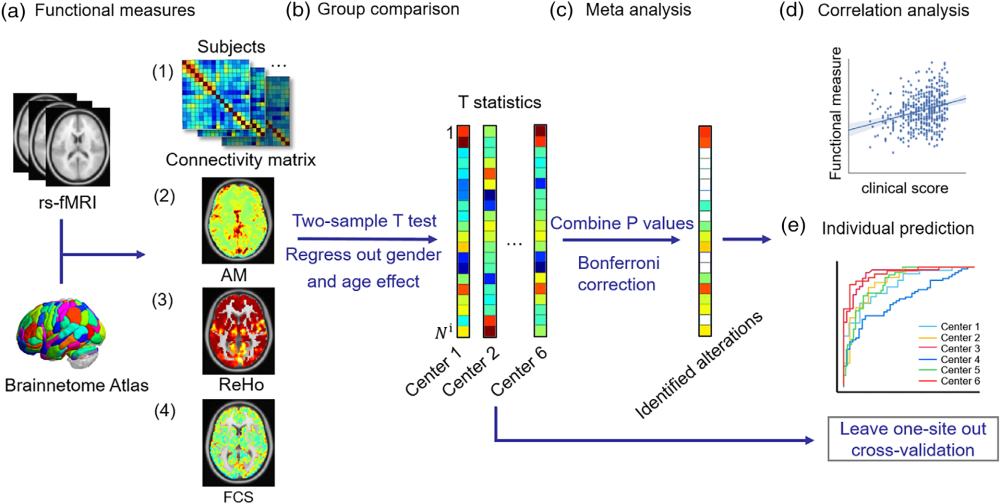 |
Figure. Schematicofthedataanalysispipeline.(a)Functionalmeasures(AM,ReHo,FCS)andtheconnectivitymatrixarecalculatedbased onBrainnetomeAtlas.(b)Atwo-samplettestwasperformedtoobtainthepvalueforeachfunctionalmeasureandconnectivityineachcenter aftercontrollingforageandgendereffects.(c)Themeta-analysiswasappliedtointegrateresultsfromsixcenters,andthesignificantlyaltered regionswereidentifiedaftermultiplecomparisoncorrection.(d)Then,thecorrelationanalysiswasperformedtoevaluatetherelationship betweenfunctionalmeasuresandtheclinicalscores.(e)Finally,leave-one-site-outcross-validationwasperformed.AM,theamplitudeoflocal brainactivity;FCS,functionalconnectivitystrength;ReHo,regionalhomogeneity |
